TiO2 Nanoparticles-Graphene Composite for Photocatalytic Application
Main Article Content
Abstract
Titanium dioxide (TiO2) nanoparticles, a candidate photocatalyst for organic pollutant treatment is controversially suspected from negative impact by the remaining residues. In this study, TiO2- graphene composite pellet was proposed as a removable catalyst for wastewater treating. Graphene was selected to enhance photocatalytic activity and promote the adhesion of composite ceramic. TiO2-graphene composite pellet was successfully fabricated by solvothermal synthesis, mixing, hydraulic compressing and sintering which were characterized by X-ray diffracting (XRD) analysis, Fourier transform infrared spectroscopy (FT-IR) and scanning electron microscopy (SEM). Preliminary test of photocatalytic activity was evaluated by determining methylene blue (MB) degradation efficiency where 74% of MB was degraded within 2 hrs in the visible region (λmax at 662 nm). Furthermore, nanocomposite pellet can be easily retrieved without degradation in solution after treatment.
Article Details
References
Frank S.N. and Bard A.J., Heterogeneous photocatalytic oxidation of cyanide ion in aqueous solutions at titanium dioxide powder, Journal of the American Chemical Society, 99: 1977, 303-304.
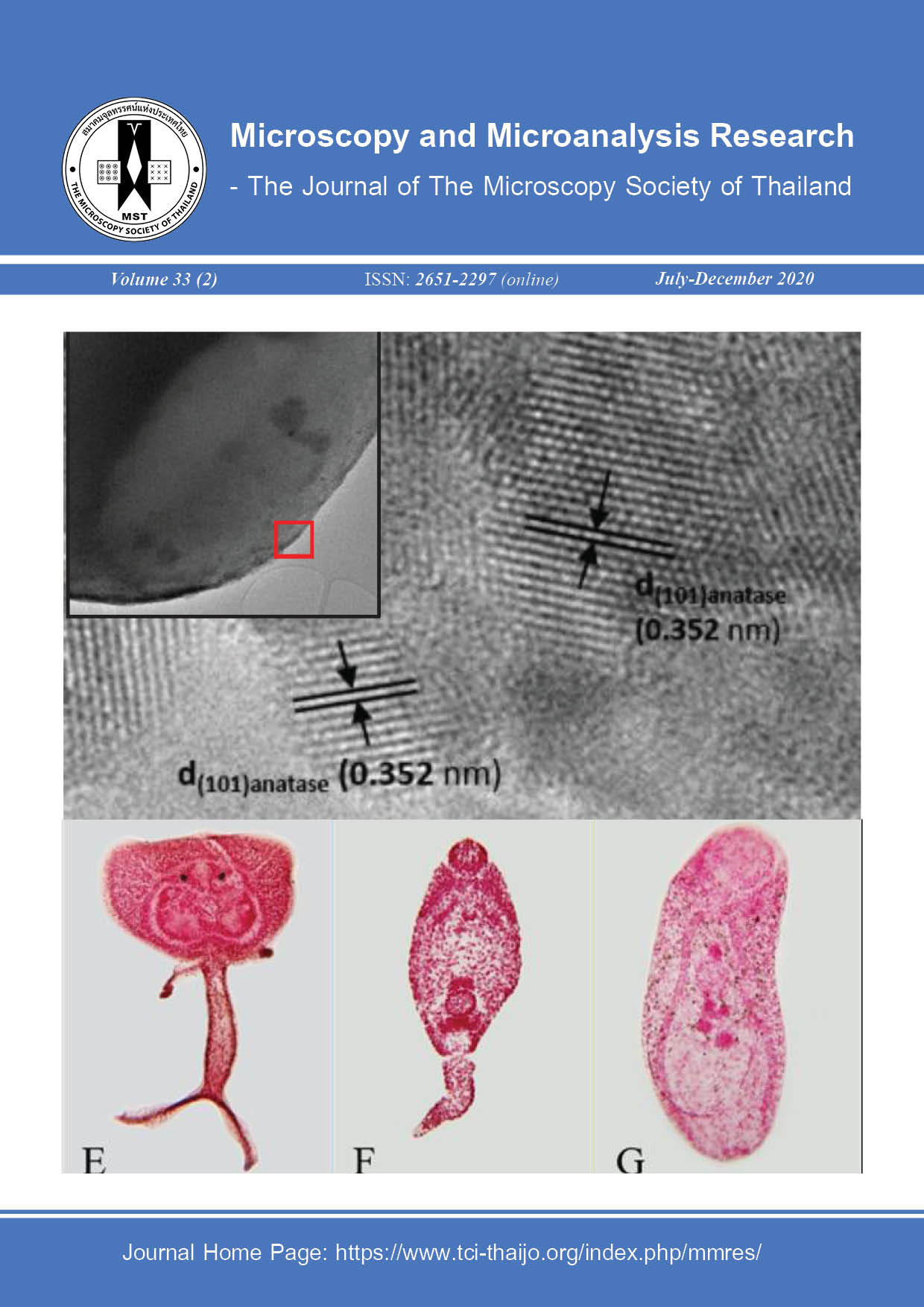
Hashimoto K., Irie H. and Fujishima A., TiO2 Photocatalysis: A Historical Overview and Future Prospects, 44: 2005, 8269-8285.
Kometani N., Fujita A. and Yonezawa Y., Synthesis of N-doped titanium oxide by hydrothermal treatment, Journal of Materials Science, 43: 2008, 2492-2498.
Daude N., Gout C. and Jouanin C., Electronic band structure of titanium dioxide, Physical Review B, 15: 1977, 3229-3235.
Irie H., Watanabe Y. and Hashimoto K., Nitrogen-Concentration Dependence on Photocatalytic Activity of TiO2-xNx Powders, The Journal of Physical Chemistry B, 107: 2003, 5483-5486.
Binas V., Venieri D., Kotzias D. and Kiriakidis G., Modified TiO2 based photocatalysts for improved air and health quality, Journal of Materiomics, 3: 2017, 3-19.
Wang Y., Zhang R., Li J., Li L. and Lin S., First-principles study on transition metal-doped anatase TiO2, Nanoscale Research Letters, 9: 2014, 46.
Hamal D.B. and Klabunde K.J., Synthesis, characterization, and visible light activity of new nanoparticle photocatalysts based on silver, carbon, and sulfur-doped TiO2, Journal of Colloid and Interface Science, 311: 2007, 514-522.
Dong H., Zeng G. and Tang L., An overview on limitations of TiO2-based particles for photocatalytic degradation of organic pollutants and the corresponding countermeasures. Water Research, 79: 2015, 128-146.
Bessekhouad Y., Robert D. and Weber J.V., Synthesis of photocatalytic TiO2 nanoparticles: optimization of the preparation conditions. Journal of Photochemistry and Photobiology A: Chemistry, 157: 2003 47-53.
Mahmoud W.M.M., Rastogi T. and Kümmerer K., Application of titanium dioxide nanoparticles as a photocatalyst for the removal of micropollutants such as pharmaceuticals from water. Current Opinion in Green and Sustainable Chemistry, 6: 2017, 1-10.
Mahvi A.H., Ghanbarian M., Nasseri S. and Khairi A., Mineralization and discoloration of textile wastewater by TiO2 nanoparticles. Desalination, 239: 2009, 309-316.
Xue H., Jiang Y. and Yuan K., Floating photocatalyst of B–N–TiO2/expanded perlite: a sol–gel synthesis with optimized mesoporous and high photocatalytic activity. Scientific Reports, 6: 2016, 29902.
Machado L.C.R., Torchia C.B. and Lago R.M., Floating photocatalysts based on TiO2 supported on high surface area exfoliated vermiculite for water decontamination. Catalysis Communications, 7: 2006, 538-541.
Magalhães F., Moura F.C.C. and Lago R.M., TiO2/LDPE composites: A new floating photocatalyst for solar degradation of organic contaminants. Desalination, 276: 2011, 266-271.
Magalhães F. and Lago R.M., Floating photocatalysts based on TiO2 grafted on expanded polystyrene beads for the solar degradation of dyes. Solar Energy, 83: 2009, 1521-1526.
Hummers W.S. and Offeman R.E., Preparation of graphitic oxide. Journal of the american chemical society, 80: 1958, 1339-1339.
Luttrell T., Halpegamage S. and Tao J., Why is anatase a better photocatalyst than rutile - Model studies on epitaxial TiO2 films. Scientific Reports, 4: 2014, 40-43.
Silverstein R.M. and Bassler G.C., Spectrometric identification of organic compounds, Journal of Chemical Education, 39: 1962, 546.
Nolan N.T., Seery M. K. and Pillai S.C., Spectroscopic Investigation of the Anatase-to-Rutile Transformation of Sol−Gel-Synthesized TiO2 Photocatalysts, The Journal of Physical Chemistry C, 113: 2009, 1651-16157.
Hamam K.A. and Gaabour L.H., Verification of the changes in the structural and physical properties of PU/PEO embedded with graphene oxide, Results in Physics, 7: 2017, 2727-2431.