Antagonistic Activity against Plant Pathogenic Fungus by Various Indigenous Microorganisms from Different Cropping Systems in Soc Trang Province, Vietnam DOI: 10.32526/ennrj.18.3.2020.23
Main Article Content
Abstract
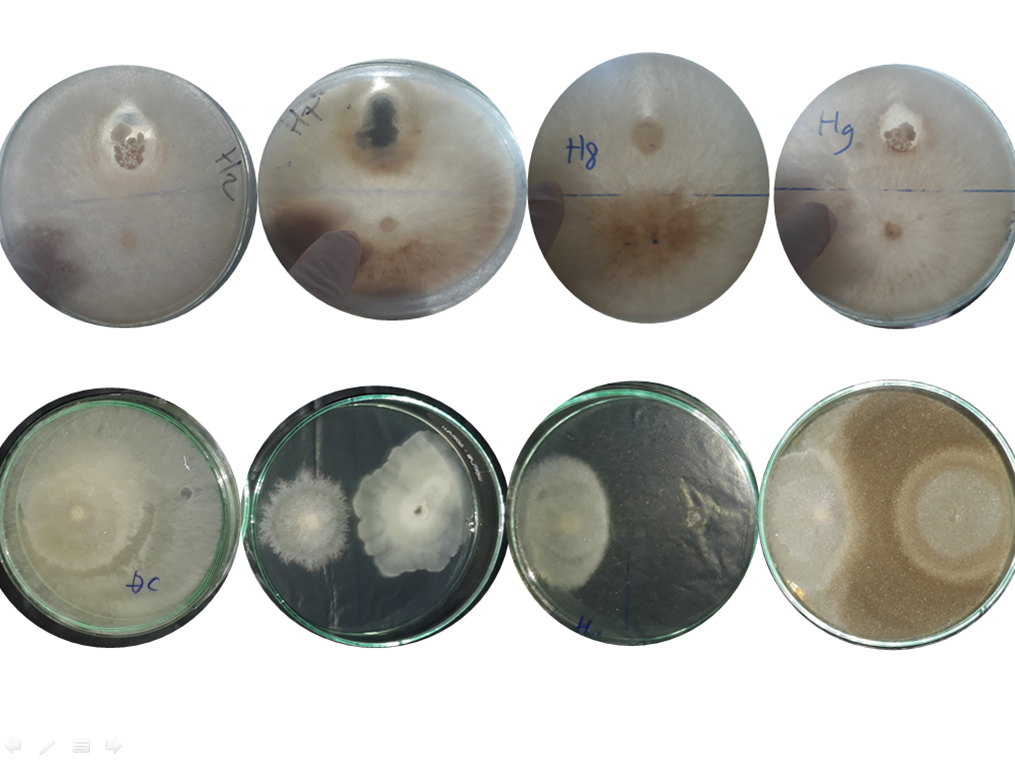
This study assessed antagonistic capacity of various indigenous microorganisms (IMO) collected from different cropping systems within Soc Trang province against plant pathogenic fungus including Fusarium oxysporum and Rhizoctonia solani. Biocontrol activity of fifteen collected IMOs was investigated on PDA agar media for 5-7 days under laboratory conditions with three different scenarios. IMO and pathogenic fungus were incubated at the same time and IMO was introduced before and after inoculation of plant pathogenic fungus. The results illustrated that all surveyed IMOs were found to have highly potential biocontrol against two plant pathogenic fungi to different extents and IMOs which were introduced before the inoculation of pathogenic fungi showed the highest efficiency in biocontrol of plant pathogen. Particularly, four out of fifteen IMOs which were collected from bamboo, shallot, grapefruit and guava farms showed their highest antagonistic efficacy on Fusarium oxysporum biocontrol by completely decaying fungal hyphae of this fungal strain after seven incubation days. For Rhizoctonia solani, all IMOs displayed highly antagonistic ability with inhibitory percentages varying between 52.96% and 92.59% after two days. The antagonistic functions of all collected IMOs could be exploited for plant protection from plant-pathogenic fungus.
Article Details
Published articles are under the copyright of the Environment and Natural Resources Journal effective when the article is accepted for publication thus granting Environment and Natural Resources Journal all rights for the work so that both parties may be protected from the consequences of unauthorized use. Partially or totally publication of an article elsewhere is possible only after the consent from the editors.
References
2. Chiemela FA, Serafin LN, Ricardo LI, Joseph LN. Isolation and characterization of indigenous microorganism (IMO) from fugao bamboo (Phyllostachys Aurea) forest. International Journal of Science and Research 2013a;4(2):1319-24.
3. Chiemela FA, Serafin LN, Ricardo LI, Joseph LN. Application of indigenous microorganisms (IMO) for bio-conversion of agricultural waste. International Journal of Science and Research 2013b;4(5):778-84.
4. Dar WA, Beig MA, Ganie SA, Bhat JA, Shabir-u-Rehman, Razvi SM. In vitro study of fungicides and biocontrol agents against Fusarium oxysporum f.sp. pini causing root rot of Western Himalayan fir (Abies pindrow). Academin Journal: Scientific Research and Essays 2013;8(30):1407-12.
5. Duffy BK, Weller DM. Use of Gaeumannomyces graminis var. graminis alone and in combination with fluorescent Pseudomonas spp. to suppress take-all of wheat. Plant Disease 1995;79:907-11.
6. Fierer N, Lauber CL, Ramirez KS, Zaneveld J, Bradford MA, Knight R. Comparative metagenomic, phylogenetic and physiological analyses of soil microbial communities across nitrogen gradients. International Society for Microbial Ecology Journal 2012;6:1007-17.
7. Ghai S, Sood SS, Jain RK. Antagonistic and antimicrobial activities of some bacterial isolates collected from soil samples. Indian Journal of Microbiology 2007;47:77-80.
8. Hervas A, Landa B, Datnoff LEm, Jime´nez-Dı´az RM. Effects of commercial and indigenous microorganisms on Fusarium wilt development in chickpea. Biological Control 1998;13(3):166-76.
9. Huy ND, Nguyen PQ, Hong NTT, Giang H, Vien NV, Canh NT. Isolation and evaluation of antagonistic ability of Trichoderma asperellum against soil borne plant pathogen. Vietnam Journal of Agricultural Sciences 2017;15(12):1593-640.
10. Koche D, Gade RM, Deshmukh AG. Antifungal activity of secondary metabolites produced by Pseudomonas fluorescens. The Bioscan 2013;8(2):723-6.
11. Kumar BL, Gopal DVR. Effective role of indigenous microorganism for sustainable environment. 3 Biotech 2015; 5(6):867-76.
12. Kyu CH, Koyama A. Korean Natural Farming: Indigenous Microorganisms and Vital Power of Crop/Livestock. Republic of Korea: Korean Nature Farming Association Publisher: 1997. p. 39-48.
13. Ramzan N, Noreen N, Shahzad S. Inhibition of in vitro growth of soil-borne pathogens by compost-inhabiting indigenous bacteria and fungi. Pakistan Journal of Botany 2014; 46(3):1093-9.
14. Reddy R. Cho’s Global Natural Farming. Bengaluru, India: South Asia Rural Reconstruction Association; 2011.
15. Robles-Yerena L, Rodríguez-Villarreal RA, Ortega-Amaro MA, Fraire-elázquez S, Simpson J, Rodríguez-Guerra R, Jiménez-Bremont JF. Characterization of a new fungal antagonist of Phytophthora capsici. Scientia Horticulturae 2010; 125(3):248-55.
16. Stanojković-Sebić A, Pavlović S, Starović M, Pivić R, Dinić1 Z, Lepšanović Z, Jošić D. Antagonistic activity of indigenous Pseudomonas isolates against Fusarium species isolated from anise. Scientific Papers. Series B. Horticulture 2017;61:413-6.
17. Toppo SR, Naik UC. Isolation and characterization of bacterial antagonist to plant pathogenic fungi (Fusarium spp.) from agro based area of Bilaspur. International Journal of Research Studies in Biosciences 2015:6-14.
18. Vincent JM. Distortion of fungal hyphae in the presence of certain inhibitors. Nature 1947;159(4051):850.
19. Xa LT, Nghia NK. Microbial diversity of indigenous microorganism communities from different agri-ecosystems in Soc Trang province, Vietnam. Proceeding of the International Conference on Biotechnology of Ho Chi Minh City Open University 2019: Research and Application in Biotechnology; 2019 July 26; Ho Chi Minh City Open University, Vietnam; 2019.
20. Yuliar S, Supriyati D, Rahmansyah M. Biodiversity of endophytic bacteria and their antagonistic activity to rhizoctonia solaniand Fusarium oxysporum. Global Journal of Biology, Agriculture and Health Sciences 2013;2(4):111-8.
21. Zhang C, Liu G, Xue S, Wang G. Soil bacterial community dynamics reflect changes in plant community and soil properties during the secondary succession of abandoned farmland in the Loess Plateau. Soil Biology and Biochemistry 2016;97:40-9.