Toxicological Effects of Tributyltin in Zebrafish (Danio rerio) Embryos 10.32526/ennrj/20/202200001
Main Article Content
Abstract
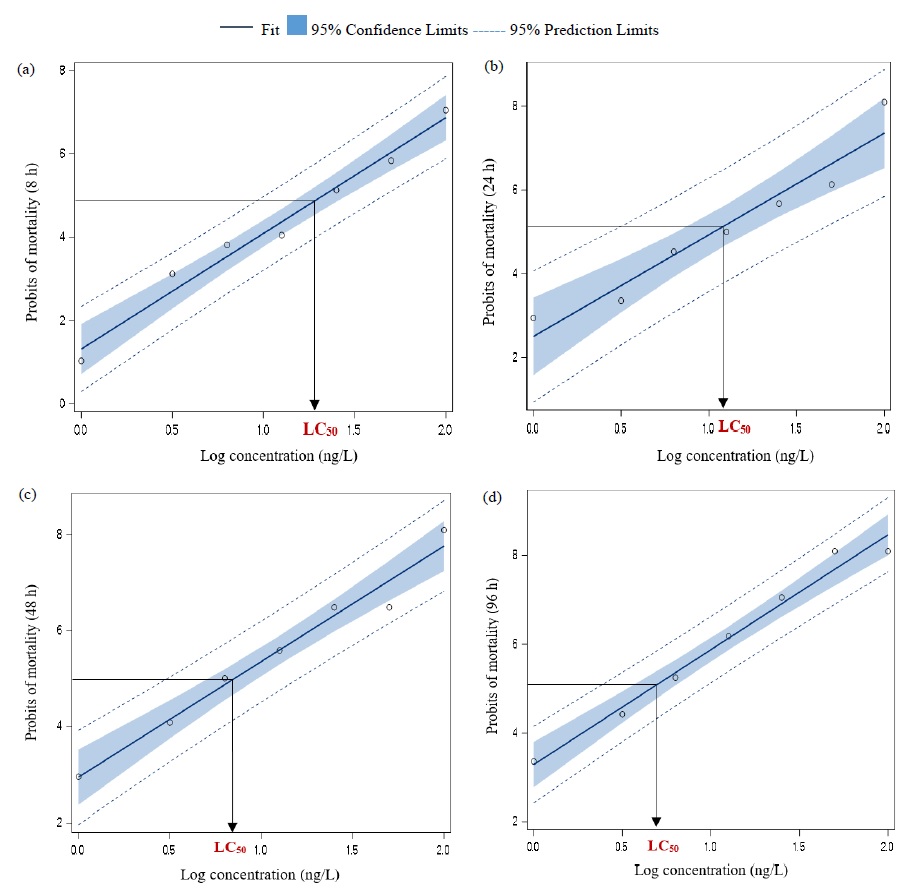
Tributyltin (TBT) is known as an endocrine-disrupting chemical abundant in the aquatic environment. In the present study, zebrafish fish embryos were used to observe the chronic toxicity of TBT. Fish embryo toxicity analysis was carried out for different TBT concentrations (100, 50, 25, 12.5, 6.2, and 3.1 ng/L) and fertilized eggs were used to test each concentration effect. Fertilized eggs in 24-well plates (20 eggs in each well) were incubated at 26°C for four days and embryo coagulation, heartbeat of the embryo and mortality lethal endpoints (LC50 values) were recorded after 8, 24, 48, and 96 h. The results revealed that 100% coagulations of the embryos occurred at TBT doses of 50 and 100 ng/L. The coagulation significantly increased in a dose-dependent manner and TBT might induce coagulation of zebrafish embryos. Heartbeat changes were significantly decreased (p<0.05) in a dose-dependent manner at different TBT doses. LC50 values of TBT for zebrafish embryos were 19.9, 11.7, 7.3, and 5.2 ng/L at 8, 24, 48, and 96 h, respectively. The percentage of mortality was higher in embryos for the trace level of TBT, indicating that embryos are more sensitive to TBT toxicity. Hence, TBT is highly toxic and leads to a lethal effect on the zebrafish embryo, resulting in species extinction and declining biodiversity in the aquatic environment.
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Published articles are under the copyright of the Environment and Natural Resources Journal effective when the article is accepted for publication thus granting Environment and Natural Resources Journal all rights for the work so that both parties may be protected from the consequences of unauthorized use. Partially or totally publication of an article elsewhere is possible only after the consent from the editors.
References
Ain QU, Roy D, Ahsan A, Farooq MA, Aquib M, Hussain Z, et al. Endocrine-disrupting chemicals: Occurrence and exposure to the human being. In: Akash MSH, Rehman K, Hashmi MZ, editors. Endocrine Disrupting Chemicals-induced Metabolic Disorders and Treatment Strategies. Emerging Contaminants and Associated Treatment Technologies. Switzerland: Springe Cham; 2021. p. 113-23.
Bandara KRV, Chinthaka SDM, Yasawardene SG, Manage PM. Modified, optimized method of determination of Tributyltin (TBT) contamination in coastal water, sediment and biota in Sri Lanka. Marine Pollution Bulletin 2021;166:Article No. 112202.
Braunbeck T, Bottcher M, Hollert H, Kosmehl T, Lammer E, Leist E, et al. Towards an alternative for the acute fish LC50 test in chemical assessment: The fish embryo toxicity test goes multi-species-an update. ALTEX-Alternatives to Animal Experi-mentation 2005;22:87-102.
Bushong SJ, Hall JLW, Hall WS, Johnson WE, Herman RL. Acute toxicity of tributyltin to selected Chesapeake Bay fish and invertebrates. Water Research 1988;22:1027-32.
Corsi I, Bergami E, Grassi G. Behavior and bio-interactions of anthropogenic particles in marine environment for a more realistic ecological risk assessment. Frontiers in Envi-ronmental Science 2020;8:1-21.
Cui Z, Zhang K, Zhou Q, Liu J, Jiang G. Determination of methyltin compounds in urine of occupationally exposed and general population by in situ ethylation and headspace SPME coupled with GC-FPD. Talanta 2011;85:1028-33.
De Oliveira GAR, Leme DM, de Lapuente J, Brito LB, Porredon C, de Brito Rodrigues L, et al. A test battery for assessing the ecotoxic effects of textile dyes. Chemico-Biological Interactions 2018;291:171-9.
Dimitriou P, Castritsi-Catharios J, Miliou H. Acute toxicity effects of tributyltin chloride and triphenyltin chloride on gilthead seabream, Sparus aurata L., embryos. Ecotoxicology and Environmental Safety 2003;54:30-5.
Fent K. Ecotoxicological effects at contaminated sites. Toxicology 2004;205:223-40.
Griffin MD, Pereira SR, DeBari MK, Abbott RD. Mechanisms of action, chemical characteristics, and model systems of obesogens. BMC Biomedical Engineering 2020;2:1-13.
Guruge KS, Manage PM, Yamanaka N, Miyazaki S, Taniyasu S, Yamashita N. Species-specific concentrations of perfluoroalkyl contaminants in farm and pet animals in Japan. Chemosphere 2008;73:210-5.
He S, Li P, Li ZH. Review on endocrine disrupting toxicity of triphenyltin from the perspective of species evolution: Aquatic, amphibious and mammalian. Chemosphere 2020; 269:Article No. 128711.
International Marine Organisation (IMO). International convention on the control of harmful antifouling systems on ships [Internet]. 2001 [cited 2021 Mar 21]. Available from: https://www.imo.org/en/About/Conventions/Pages/International-Convention-on-the-Control-of-Harmful-Anti-fouling-Systems-on-Ships-(AFS).aspx.
Kim BM, Saravanan M, Lee DH, Kang JH, Kim M, Jung JH, et al. Exposure to sublethal concentrations of tributyltin reduced survival, growth, and 20-hydroxyecdysone levels in a marine mysid. Marine Environmental Research 2018;140:96-103.
Lammer E, Carr GJ, Wendler K, Rawlings JM, Belanger SE, Braunbeck T. Is the fish embryo toxicity test (FET) with the zebrafish (Danio rerio) a potential alternative for the fish acute toxicity test? Comparative Biochemistry and Physiology Part C: Toxicology and Pharmacology 2009;149:196‐209.
Langston WJ. Endocrine disruption and altered sexual development in aquatic organisms: An invertebrate perspective. Journal of the Marine Biological Association of the United Kingdom 2020;100:495-515.
Lu Y, Zhang Y, Zhong C, Martin JW, Alessi DS, Goss GG, et al. Suspended solids-associated toxicity of hydraulic fracturing flowback and produced water on early life stages of zebrafish (Danio rerio). Environmental Pollution 2021;287:Article No. 117614.
Lungu-Mitea S, Vogs C, Carlsson G, Montag M, Frieberg, K, Oskarsson A, et al. Modeling bioavailable concentrations in zebrafish cell lines and embryos increases the correlation of toxicity potencies across test systems. Environmental Science and Technology 2020;55:447-57.
Lv H, Wang J, Wang M, Shen L, Xiao L, Chen T, et al. Potent inhibition of tributyltin (TBT) and triphenyltin (TPT) against multiple UDP-glucuronosyltransferases (UGT): A new potential mechanism underlying endocrine disrupting actions. Food and Chemical Toxicology 2021;149:Article No. 112039.
Mendis JC, Tennakoon TK, Jayasinghe CD. Zebrafish embryo toxicity of a binary mixture of pyrethroid insecticides: d-tetramethrin and cyphenothrin. Journal of Toxicology 2018; 2018:1-8.
Ohura T, Sakakibara H, Watanabe I, Shim WJ, Manage PM, Guruge KS. Spatial and vertical distributions of sedimentary halogenated polycyclic aromatic hydrocarbons in moderately polluted areas of Asia. Environmental Pollution 2015; 196:331-40.
Pinkney AE, Matteson LL, Wright DA. Effects of tributyltin on survival, growth, morphometry, and RNA-DNA ratio of larval striped bass, Morone saxatilis. Archives of Environmental Contamination and Toxicology 1990;19:235-40.
Santos D, Vieira R, Luzio A, Felix L. Zebrafish early life stages for toxicological screening: Insights from molecular and biochemical markers. Advances in Molecular Toxicology 2018;12:151-79.
Scholz S, Fischer S, Gundel U, Küster E, Luckenbach T, Voelker D. The zebrafish embryo model in environmental risk assessment: Applications beyond acute toxicity testing. Environmental Science and Pollution Research 2008;15: 394-404.
Sekizawa J, Sutur G, Birnbaum L. Integrated human and ecological risk assessment: A case study of tributyltin and triphenyltin compounds. Human and Ecological Risk Assessment 2003;9:325-42.
Sousa AC, Pastorinho MR, Takahashi S, Tanabe S. History on organotin compounds, from snails to humans. Environmental Chemistry Letters 2014;12(1):117-37.
Strand J, Jacobsen JA, Pedersen B, Granmo A. Butyltin compounds in sediment and molluscs from the shipping strait between Denmark and Sweden. Environmental Pollution 2003;124:7-15.
Thitinarongwate W, Mektrirat R, Nimlamool W, Khonsung P, Pikulkaew S, Okonogi S, et al. Phytochemical and safety evaluations of Zingiber ottensii valeton essential oil in zebrafish embryos and rats. Toxics 2021;9:102-12.
Xiao X, Zhu S, Zou X, He G, Jiang J, Sheng GD. Relation of tributyltin and triphenyltin equilibrium sorption and kinetic accumulation in carp and Ceratophyllum demersum. Ecotoxicology and Environmental Safety 2020;192:Article No. 110289.