Carbohydrate Interfaces in Plant-Fungal Interactions: A Scoping Review
Main Article Content
Abstract
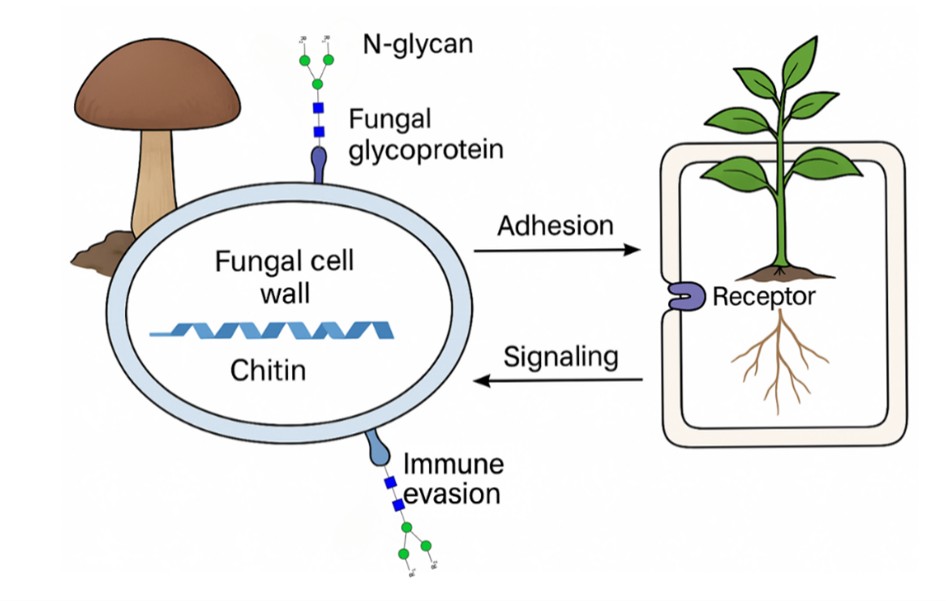
Carbohydrates play important roles in plant–fungal interactions, contributing to fungal adhesion, immune evasion, and host recognition. During fungal infection in plants, carbohydrate-based structures play a significant role in fungal adhesion, immune evasion, and host recognition. This scoping review aims to consolidate current evidence on the role of carbohydrate interfaces, including both structural polysaccharides and protein-linked carbohydrates, in plant–fungal interactions. This study followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Extension for Scoping Reviews guidelines. A total of 451 articles were retrieved from Scopus, Web of Science, PubMed, and ScienceDirect using keywords such as “carbohydrate”, “fungal”, “glycan”, “infection”, and “plant”, from which 20 articles were selected for analysis based on inclusion criteria. Chitin-related glycans were the most frequently investigated carbohydrate type (n = 10), followed by N-glycans (n = 4) and β-glucans (n = 4), particularly in major crop systems. In contrast, protein-linked carbohydrates, especially N-glycans, remain limited, while studies specifically addressing O-glycans are largely absent. Most research has also focused on staple crops and laboratory-based experimental systems. These gaps suggest the need for greater focus on functional and dynamic analyses of glycan modifications during infection, broader representation of crops, and the application of advanced glycomic and molecular approaches.
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
References
Trouvelot, S.; Héloir, M.-C.; Poinssot, B.; Gauthier, A.; Paris, F.; Guillier, C.; Combier, M.; Trdá, L.; Daire, X.; Adrian, M. Carbohydrates in Plant Immunity and Plant Protection: Roles and Potential Application as Foliar Sprays. Front. Plant Sci., 2014, 5. https://doi.org/10.3389/fpls.2014.00592
Ramaprabha, K.; Venkat Kumar, S.; Saravanan, P.; Rajeshkannan, R.; Rajasimman, M.; Kamyab, H.; Vasseghian, Y. Exploring the Diverse Applications of Carbohydrate Macromolecules in Food, Pharmaceutical, and Environmental Technologies. Environ. Res., 2024, 240, 117521. https://doi.org/10.1016/j.envres.2023.117521
Poole, C. F. Application of Thin-Layer Chromatography to the Analysis of Saccharides. In Instrumental Thin-Layer Chromatography; Elsevier, 2023; pp 413–435. https://doi.org/10.1016/B978-0-323-99970-0.00020-X
Bushra, R.; Ahmad, M.; Seidi, F.; Qurtulen; Song, J.; Jin, Y.; Xiao, H. Polysaccharide-Based Nanoassemblies: From Synthesis Methodologies and Industrial Applications to Future Prospects. Adv. Colloid Interface Sci., 2023, 318, 102953. https://doi.org/10.1016/j.cis.2023.102953
Han, Z.; Schneiter, R. Dual Functionality of Pathogenesis-Related Proteins: Defensive Role in Plants versus Immunosuppressive Role in Pathogens. Front. Plant Sci., 2024, 15. https://doi.org/10.3389/fpls.2024.1368467
Nishad, R.; Ahmed, T.; Rahman, V. J.; Kareem, A. Modulation of Plant Defense System in Response to Microbial Interactions. Front. Microbiol., 2020, 11. https://doi.org/10.3389/fmicb.2020.01298
Ling, L.-Z.; Chen, L.-L.; Liu, Z.-Z.; Luo, L.-Y.; Tai, S.-H.; Zhang, S.-D. Genome Sequencing and CAZymes Repertoire Analysis of Diaporthe eres P3-1W Causing Postharvest Fruit Rot of ‘Hongyang’ Kiwifruit in China. PeerJ, 2024, 12, e17715. https://doi.org/10.7717/peerj.17715
Weiland, P.; Dempwolff, F.; Steinchen, W.; Freibert, S.; Tian, H.; Glatter, T.; Martin, R.; Thomma, B. P. H. J.; Bange, G.; Altegoer, F. Structural and Functional Analysis of the Cerato‐platanin‐like Protein Cpl1 Suggests Diverging Functions in Smut Fungi. Mol. Plant Pathol., 2023, 24(7), 768–787. https://doi.org/10.1111/mpp.13349
Singh, L.; Sinha, A.; Gupta, M.; Xiao, S.; Hammond, R.; Rawat, N. Wheat Pore-Forming Toxin-like Protein Confers Broad-Spectrum Resistance to Fungal Pathogens in Arabidopsis. Molecular Plant-Microbe Interactions®, 2023, 36 (8), 489–501. https://doi.org/10.1094/MPMI-12-22-0247-R
Martinez-D’Alto, A.; Yan, X.; Detomasi, T. C.; Sayler, R. I.; Thomas, W. C.; Talbot, N. J.; Marletta, M. A. Characterization of a Unique Polysaccharide Monooxygenase from the Plant Pathogen Magnaporthe oryzae. Proceedings of the National Academy of Sciences, 2023, 120(8). https://doi.org/10.1073/pnas.2215426120
Dowd, P. F.; Naumann, T. A.; Johnson, E. T. A Maize Gene Coding for a Chimeric Superlectin Reduces Growth of Maize Fungal Pathogens and Insect Pests When Expressed Transgenically in Maize Callus. Plant Gene, 2022, 30, 100359. https://doi.org/10.1016/j.plgene.2022.100359
Wawra, S.; Fesel, P.; Widmer, H.; Neumann, U.; Lahrmann, U.; Becker, S.; Hehemann, J.; Langen, G.; Zuccaro, A. FGB1 and WSC3 Are in Planta‐ Induced β ‐glucan‐binding Fungal Lectins with Different Functions. New Phytologist, 2019, 222(3), 1493–1506. https://doi.org/10.1111/nph.15711
Hurlburt, N. K.; Chen, L.-H.; Stergiopoulos, I.; Fisher, A. J. Structure of the Cladosporium fulvum Avr4 Effector in Complex with (GlcNAc)6 Reveals the Ligand-Binding Mechanism and Uncouples Its Intrinsic Function from Recognition by the Cf-4 Resistance Protein. PLoS Pathog., 2018, 14(8), e1007263. https://doi.org/10.1371/journal.ppat.1007263
Samalova, M.; Mélida, H.; Vilaplana, F.; Bulone, V.; Soanes, D. M.; Talbot, N. J.; Gurr, S. J. The Β‐1,3‐glucanosyltransferases (Gels) Affect the Structure of the Rice Blast Fungal Cell Wall during Appressorium‐mediated Plant Infection. Cell. Microbiol., 2017, 19(3). https://doi.org/10.1111/cmi.12659
Liu, Z.; Gao, Y.; Kim, Y. M.; Faris, J. D.; Shelver, W. L.; de Wit, P. J. G. M.; Xu, S. S.; Friesen, T. L. SnTox1, a Parastagonospora nodorum Necrotrophic Effector, Is a Dual‐function Protein That Facilitates Infection While Protecting from Wheat‐produced Chitinases. New Phytologist, 2016, 211(3), 1052–1064. https://doi.org/10.1111/nph.13959
Koharudin, L. M. I.; Debiec, K. T.; Gronenborn, A. M. Structural Insight into Fungal Cell Wall Recognition by a CVNH Protein with a Single LysM Domain. Structure, 2015, 23(11), 2143–2154. https://doi.org/10.1016/j.str.2015.07.023
Chen, X.-L.; Shi, T.; Yang, J.; Shi, W.; Gao, X.; Chen, D.; Xu, X.; Xu, J.-R.; Talbot, N. J.; Peng, Y.-L. N -Glycosylation of Effector Proteins by an α-1,3-Mannosyltransferase Is Required for the Rice Blast Fungus to Evade Host Innate Immunity. Plant Cell, 2014, 26(3), 1360–1376. https://doi.org/10.1105/tpc.114.123588
Lowe, R. G. T.; Cassin, A.; Grandaubert, J.; Clark, B. L.; Van de Wouw, A. P.; Rouxel, T.; Howlett, B. J. Genomes and Transcriptomes of Partners in Plant-Fungal- Interactions between Canola (BrassicanNapus) and Two Leptosphaeria Species. PLoS One, 2014, 9(7), e103098. https://doi.org/10.1371/journal.pone.0103098
Chowdhury, J.; Henderson, M.; Schweizer, P.; Burton, R. A.; Fincher, G. B.; Little, A. Differential Accumulation of Callose, Arabinoxylan and Cellulose in Nonpenetrated versus Penetrated Papillae on Leaves of Barley Infected with Blumeria graminis F. sp. Hordei. New Phytologist, 2014, 204(3), 650–660. https://doi.org/10.1111/nph.12974
Gifoni, J. M.; Oliveira, J. T. A.; Oliveira, H. D.; Batista, A. B.; Pereira, M. L.; Gomes, A. S.; Oliveira, H. P.; Grangeiro, T. B.; Vasconcelos, I. M. A Novel Chitin‐binding Protein from Moringa Oleifera Seed with Potential for Plant Disease Control. Peptide Science, 2012, 98(4), 406–415. https://doi.org/10.1002/bip.22068
Koharudin, L. M. I.; Viscomi, A. R.; Montanini, B.; Kershaw, M. J.; Talbot, N. J.; Ottonello, S.; Gronenborn, A. M. Structure-Function Analysis of a CVNH-LysM Lectin Expressed during Plant Infection by the Rice Blast Fungus Magnaporthe oryzae. Structure, 2011, 19(5), 662–674. https://doi.org/10.1016/j.str.2011.03.004
Zhao, J.; Buchwaldt, L.; Rimmer, S. R.; Sharpe, A.; Mcgregor, L.; Bekkaoui, D.; Hegedus, D. Patterns of Differential Gene Expression in Brassica napus Cultivars Infected with Sclerotinia sclerotiorum. Mol. Plant Pathol., 2009, 10(5), 635–649. https://doi.org/10.1111/j.1364-3703.2009.00558.x
Bolton, M. D.; Van Esse, H. P.; Vossen, J. H.; De Jonge, R.; Stergiopoulos, I.; Stulemeijer, I. J. E.; Van Den Berg, G. C. M.; Borrás‐Hidalgo, O.; Dekker, H. L.; De Koster, C. G.; et al. The Novel Cladosporium fulvum Lysin Motif Effector Ecp6 Is a Virulence Factor with Orthologues in Other Fungal Species. Mol. Microbiol., 2008, 69(1), 119–136. https://doi.org/10.1111/j.1365-2958.2008.06270.x
Miya, A.; Albert, P.; Shinya, T.; Desaki, Y.; Ichimura, K.; Shirasu, K.; Narusaka, Y.; Kawakami, N.; Kaku, H.; Shibuya, N. CERK1, a LysM Receptor Kinase, Is Essential for Chitin Elicitor Signaling in Arabidopsis. Proceedings of the National Academy of Sciences, 2007, 104(49), 19613–19618. https://doi.org/10.1073/pnas.0705147104
van den Burg, H. A.; Harrison, S. J.; Joosten, M. H. A. J.; Vervoort, J.; de Wit, P. J. G. M. Cladosporium fulvum Avr4 Protects Fungal Cell Walls Against Hydrolysis by Plant Chitinases Accumulating During Infection. Molecular Plant-Microbe Interactions®, 2006, 19(12), 1420–1430. https://doi.org/10.1094/MPMI-19-1420
Dean, R. A.; Talbot, N. J.; Ebbole, D. J.; Farman, M. L.; Mitchell, T. K.; Orbach, M. J.; Thon, M.; Kulkarni, R.; Xu, J.-R.; Pan, H.; et al. The Genome Sequence of the Rice Blast Fungus Magnaporthe grisea. Nature, 2005, 434(7036), 980–986. https://doi.org/10.1038/nature03449
Deimel, L. P.; Xue, X.; Sattentau, Q. J. Glycans in HIV-1 Vaccine Design – Engaging the Shield. Trends Microbiol., 2022, 30(9), 866–881. https://doi.org/10.1016/j.tim.2022.02.004
Ha, T. K.; Kim, D.; Kim, C. L.; Grav, L. M.; Lee, G. M. Factors Affecting the Quality of Therapeutic Proteins in Recombinant Chinese Hamster Ovary Cell Culture. Biotechnol. Adv., 2022, 54, 107831. https://doi.org/10.1016/j.biotechadv.2021.107831
Fernando, L. D.; Zhao, W.; Gautam, I.; Ankur, A.; Wang, T. Polysaccharide Assemblies in Fungal and Plant Cell Walls Explored by Solid-State NMR. Structure, 2023, 31(11), 1375–1385. https://doi.org/10.1016/j.str.2023.07.012
Bartetzko, M. P.; Pfrengle, F. Automated Glycan Assembly of Plant Oligosaccharides and Their Application in Cell‐Wall Biology. ChemBioChem, 2019, 20(7), 877–885. https://doi.org/10.1002/cbic.201800641
Polko, J. K.; Kieber, J. J. The Regulation of Cellulose Biosynthesis in Plants. Plant Cell, 2019, 31(2), 282–296. https://doi.org/10.1105/tpc.18.00760
Mendu, L.; Ulloa, M.; Payton, P.; Monclova-Santana, C.; Chagoya, J.; Mendu, V. Lignin and Cellulose Content Differences in Roots of Different Cotton Cultivars Associated with Different Levels of Fusarium Wilt Race 4 (FOV4) Resistance-Response. J. Agric. Food Res., 2022, 10, 100420. https://doi.org/10.1016/j.jafr.2022.100420
Wiesinger, P.; Nestor, G. NMR Spectroscopic Studies of Chitin Oligomers – Resolution of Individual Residues and Characterization of Minor Amide Cis Conformations. Carbohydr. Polym., 2025, 351, 123122. https://doi.org/10.1016/j.carbpol.2024.123122
Mason, K. N.; Ekanayake, G.; Heese, A. Staining and Automated Image Quantification of Callose in Arabidopsis Cotyledons and Leaves; 2020; pp 181–199. https://doi.org/10.1016/bs.mcb.2020.05.005
Hayashi, T.; Kaida, R. Functions of Xyloglucan in Plant Cells. Mol. Plant, 2011, 4(1), 17–24. https://doi.org/10.1093/mp/ssq063
Wan, J.; He, M.; Hou, Q.; Zou, L.; Yang, Y.; Wei, Y.; Chen, X. Cell Wall Associated Immunity in Plants. Stress Biology, 2021, 1(1), 3. https://doi.org/10.1007/s44154-021-00003-4
Wang, D.; Lu, Q.; Jin, S.; Fan, X.; Ling, H. Pectin, Lignin and Disease Resistance in Brassica napus L.: An Update. Horticulturae, 2023, 9(1), 112. https://doi.org/10.3390/horticulturae9010112
Mélida, H.; Bacete, L.; Ruprecht, C.; Rebaque, D.; del Hierro, I.; López, G.; Brunner, F.; Pfrengle, F.; Molina, A. Arabinoxylan-Oligosaccharides Act as Damage Associated Molecular Patterns in Plants Regulating Disease Resistance. Front. Plant Sci., 2020, 11. https://doi.org/10.3389/fpls.2020.01210
Han, B.; Baruah, K.; Cox, E.; Vanrompay, D.; Bossier, P. Structure-Functional Activity Relationship of β-Glucans from the Perspective of Immunomodulation: A Mini-Review. Front. Immunol., 2020, 11. https://doi.org/10.3389/fimmu.2020.00658
Bligh, M.; Nguyen, N.; Buck-Wiese, H.; Vidal-Melgosa, S.; Hehemann, J.-H. Structures and Functions of Algal Glycans Shape Their Capacity to Sequester Carbon in the Ocean. Curr. Opin. Chem. Biol., 2022, 71, 102204. https://doi.org/10.1016/j.cbpa.2022.102204
Karuppan Perumal, M. K.; Gandhi, D.; Rajasekaran, M. B. S.; Kudiyarasu, S.; Renuka, R. R.; Julius, A.; Samrot, A. V.; Lakshmi Narayanan, A. Inhibition of Angiogenesis Using Laminarin a Natural Polysaccharide from Brown Seaweeds – A Review. Biocatal. Agric. Biotechnol., 2023, 54, 102947. https://doi.org/10.1016/j.bcab.2023.102947
Sharma, A.; Kaur, I.; Dheer, D.; Nagpal, M.; Kumar, P.; Venkatesh, D. N.; Puri, V.; Singh, I. A Propitious Role of Marine Sourced Polysaccharides: Drug Delivery and Biomedical Applications. Carbohydr. Polym., 2023, 308, 120448. https://doi.org/10.1016/j.carbpol.2022.120448
Tziros, G. T.; Samaras, A.; Karaoglanidis, G. S. Laminarin Induces Defense Responses and Efficiently Controls Olive Leaf Spot Disease in Olive. Molecules, 2021, 26(4), 1043. https://doi.org/10.3390/molecules26041043
Zang, H.; Xie, S.; Zhu, B.; Yang, X.; Gu, C.; Hu, B.; Gao, T.; Chen, Y.; Gao, X. Mannan Oligosaccharides Trigger Multiple Defence Responses in Rice and Tobacco as a Novel Danger‐associated Molecular Pattern. Mol. Plant Pathol., 2019, 20(8), 1067–1079. https://doi.org/10.1111/mpp.12811
Liu, C.; Talbot, N. J.; Chen, X. Protein Glycosylation during Infection by Plant Pathogenic Fungi. New Phytologist, 2021, 230(4), 1329–1335. https://doi.org/10.1111/nph.17207
Molodchenkova, O. О.; Ryshchakova, O. V.; Kartuzova, T. V.; Mishchenko, L. T. Characterization of Lectins from Wheat Seedlings Infected with Fusarium graminearum and Treated by Jasmonic Acid. The Ukrainian Biochemical Journal, 2023, 95(2), 83–92. https://doi.org/10.15407/ubj95.02.083
Kanda, Y.; Shinya, T.; Wari, D.; Hojo, Y.; Fujiwara, Y.; Tsuchiya, W.; Fujimoto, Z.; Thomma, B. P. H. J.; Nishizawa, Y.; Kamakura, T.; et al. Chitin‐signaling‐dependent Responses to Insect Oral Secretions in Rice Cells Propose the Involvement of Chitooligosaccharides in Plant Defense against Herbivores. The Plant Journal, 2025, 121(1). https://doi.org/10.1111/tpj.17157
Shobade, S. O.; Zabotina, O. A.; Nilsen-Hamilton, M. Plant Root Associated Chitinases: Structures and Functions. Front. Plant Sci., 2024, 15. https://doi.org/10.3389/fpls.2024.1344142
Hariharan, G.; Prasannath, K. Recent Advances in Molecular Diagnostics of Fungal Plant Pathogens: A Mini Review. Front. Cell. Infect. Microbiol., 2021, 10. https://doi.org/10.3389/fcimb.2020.600234
Kehinde, B. A.; Panghal, A.; Garg, M. K.; Sharma, P.; Chhikara, N. Vegetable Milk as Probiotic and Prebiotic Foods; 2020; pp 115–160. https://doi.org/10.1016/bs.afnr.2020.06.003
Li, Y.-B.; Xu, R.; Liu, C.; Shen, N.; Han, L.-B.; Tang, D. Magnaporthe oryzae Fimbrin Organizes Actin Networks in the Hyphal Tip during Polar Growth and Pathogenesis. PLoS Pathog., 2020, 16(3), e1008437. https://doi.org/10.1371/journal.ppat.1008437
Wei, Y.-Y.; Liang, S.; Zhu, X.-M.; Liu, X.-H.; Lin, F.-C. Recent Advances in Effector Research of Magnaporthe oryzae. Biomolecules, 2023, 13(11), 1650. https://doi.org/10.3390/biom13111650
You, H. J.; Shim, K.-C.; Kang, I.-J.; Kim, J.-M.; Kang, S.; Lee, S. Soybean Variety Saedanbaek Confers a New Resistance Allele to Phytophthora Sojae. Plants, 2023, 12(23), 3957. https://doi.org/10.3390/plants12233957
Jones, D. A. B.; Rybak, K.; Hossain, M.; Bertazzoni, S.; Williams, A.; Tan, K.-C.; Phan, H. T. T.; Hane, J. K. Repeat-Induced Point Mutations Driving Parastagonospora Nodorum Genomic Diversity Are Balanced by Selection against Non-Synonymous Mutations. Commun. Biol., 2024, 7(1), 1614. https://doi.org/10.1038/s42003-024-07327-7
Buttar, Z. A.; Cheng, M.; Wei, P.; Zhang, Z.; Lv, C.; Zhu, C.; Ali, N. F.; Kang, G.; Wang, D.; Zhang, K. Update on the Basic Understanding of Fusarium graminearum Virulence Factors in Common Wheat Research. Plants, 2024, 13(8), 1159. https://doi.org/10.3390/plants13081159
Peng, R.; Sun, S.; Li, N.; Kong, L.; Chen, Z.; Wang, P.; Xu, L.; Wang, H.; Geng, X. Physiological and Transcriptome Profiling Revealed Defense Networks during Cladosporium fulvum and Tomato Interaction at the Early Stage. Front. Plant Sci., 2022, 13. https://doi.org/10.3389/fpls.2022.1085395
Zuo, W.; Depotter, J. R. L.; Stolze, S. C.; Nakagami, H.; Doehlemann, G. A Transcriptional Activator Effector of Ustilago maydis Regulates Hyperplasia in Maize during Pathogen-Induced Tumor Formation. Nat. Commun., 2023, 14 (1), 6722. https://doi.org/10.1038/s41467-023-42522-w
Waheed, A.; Haxim, Y.; Islam, W.; Ahmad, M.; Muhammad, M.; Alqahtani, F. M.; Hashem, M.; Salih, H.; Zhang, D. Climate Change Reshaping Plant-Fungal Interaction. Environ. Res., 2023, 238, 117282. https://doi.org/10.1016/j.envres.2023.117282
Jabiri, S.; El Hamss, H.; Amraoui, M. B.; Lahlali, R. Influence of Culture Media and Environmental Factors (Water Potential and Temperature) on Mycelial Growth of Phytopythium vexans (de Bary), the Causal Agent of Dieback Disease in Apple Trees. Appl. Microbiol., 2022, 2(4), 861–872. https://doi.org/10.3390/applmicrobiol2040066
Wang, Q.; Huang, Z.; Khan, I. A.; Li, Y.; Wang, J.; Wang, J.; Liu, X.-H.; Lin, F.; Lu, J. Key Transcription Factors Required for Outburst of Rice Blast Disease in Magnaporthe oryzae. Phytopathology Research, 2024, 6(1), 5. https://doi.org/10.1186/s42483-024-00225-0
Qiu, J.; Liu, Z.; Xie, J.; Lan, B.; Shen, Z.; Shi, H.; Lin, F.; Shen, X.; Kou, Y. Dual Impact of Ambient Humidity on the Virulence of Magnaporthe oryzae and Basal Resistance in Rice. Plant Cell Environ., 2022, 45(12), 3399–3411. https://doi.org/10.1111/pce.14452
Alam, M. S.; Maina, A. W.; Feng, Y.; Wu, L.-B.; Frei, M. Interactive Effects of Tropospheric Ozone and Blast Disease (Magnaporthe oryzae) on Different Rice Genotypes. Environmental Science and Pollution Research, 2022, 29(32), 48893–48907. https://doi.org/10.1007/s11356-022-19282-z
Liu, L.-W.; Hsieh, S.-H.; Lin, S.-J.; Wang, Y.-M.; Lin, W.-S. Rice Blast (Magnaporthe oryzae) Occurrence Prediction and the Key Factor Sensitivity Analysis by Machine Learning. Agronomy, 2021, 11(4), 771. https://doi.org/10.3390/agronomy11040771
Timmusk, S.; Nevo, E.; Ayele, F.; Noe, S.; Niinemets, Ü. Fighting Fusarium Pathogens in the Era of Climate Change: A Conceptual Approach. Pathogens, 2020, 9(6), 419. https://doi.org/10.3390/pathogens9060419