Rhodocista sp. Strain SAIYAI: A Natural Source of Spirilloxanthin and Feed Attractability in Pacific White Shrimp (Litopenaeus vannamei)
Main Article Content
Abstract
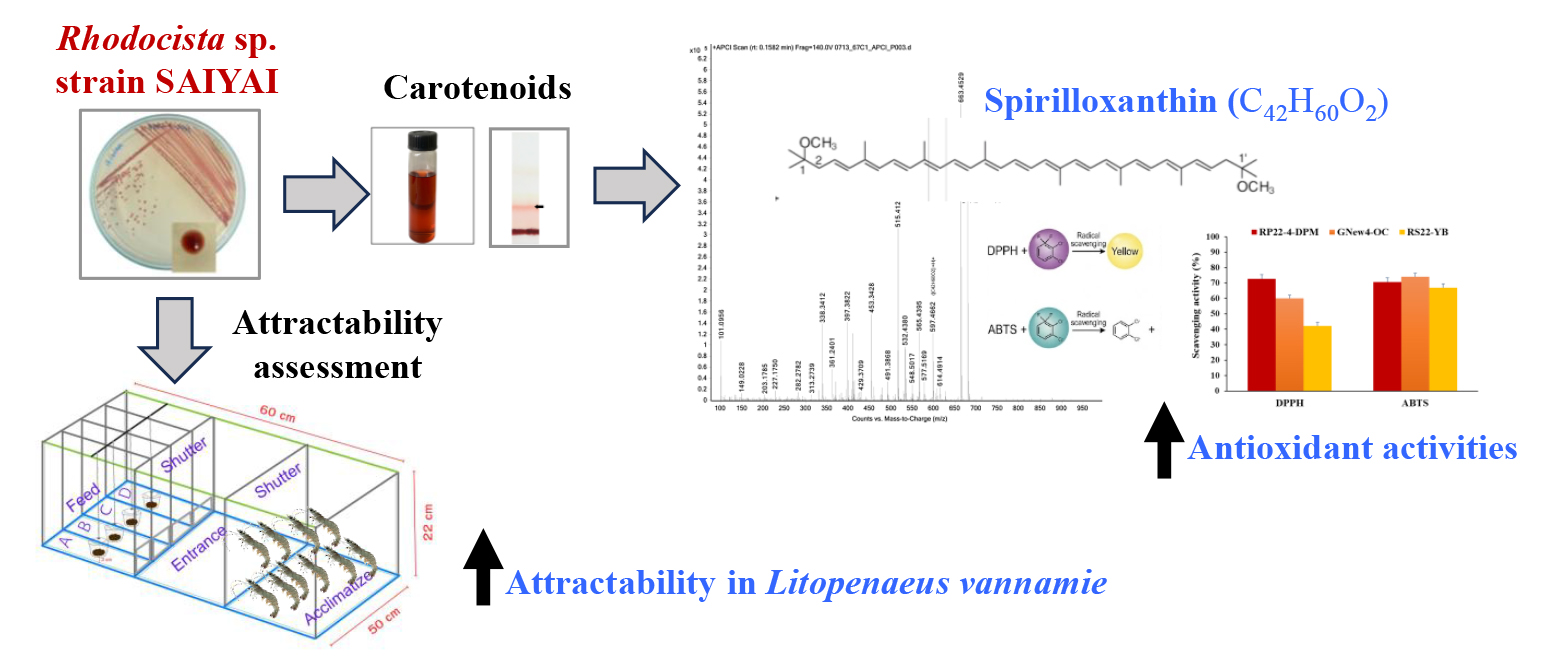
This study investigated the potential of isolated photosynthetic bacteria (PSB) as carotenoid producers and assessed their efficacy as a low-cost functional feed additive for Pacific white shrimp (Litopenaeus vannamei). Fifteen PSB isolates representing 3 color groups, red, orange, and yellow, were evaluated for total carotenoid content (TCC), and the highest TCC-producing PSB were RP22-4-DPM with 0.82 ± 0.07 mg/g of DW, RP22-OR with 0.38 ± 0.03 mg/g of DW, and RS22-YB with 0.47 ± 0.07 mg/g of DW, respectively. Isolate RP22-4-DPM, further designated as PSB strain SAIYAI, exhibited the highest DPPH scavenging activity (72.65 ± 4.60%) (p<0.05); meanwhile, its ABTS scavenging activity (70.7 ± 2.73%) showed no significant difference compared to other strains (p>0.05). The predominant pigment and the major carotenoid of this strain were identified as spirilloxanthin by UV-Vis spectrophotometry and APCI LC/MS. Additionally, the attractability was evaluated by coating soybean meal with the lyophilized cell and culture media of the SAIYAI strain at 0.5%, 1%, and 1.5% to Pacific white shrimp. All concentrations significantly increased feed attractability compared to non-coated soybean meal (p<0.05), while 0.5% and 1.0% exhibited similar attractability (p>0.05). Molecular identification through 16S rRNA sequence analysis and phylogenetic tree construction suggested that strain SAIYAI belongs to the genus Rhodocista, which is closely related to Rhodocista xerospirillum and Rhodospirillum centenum. Accordingly, this study provides evidence that Rhodocista sp. strain SAIYAI is a potent spirilloxanthin producer with antioxidant properties and potential as a multifunctional, cost-effective attractant in shrimp feed.
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
References
Kato, S.; Panitchat, S.; Boonming, S.; Teratnatorn, V.; Saito, N.; Kojima, T.; Matsui, T.; Thanasukarn, P.; Chantrapromma, K.; Aksornkoae, S. Rehabilitation of Abandoned Shrimp Ponds through Mangrove Planting at Nakhon Si Thammarat, Southern Thailand: Investigation of a Food Chain System at a Newly Developed Mangrove Ecosystem. Walailak J. Sci. Technol. 2011, 5, 137–149.
Yadav, S.; Goyal, V. C. Current Status of Ponds in India: A Framework for Restoration, Policies and Circular Economy. Wetlands 2022, 42, 107. https://doi.org/10.1007/s13157-022-01624-9
Hallenbeck, P. C.; Benemann, J. R. Biological Hydrogen Production: Fundamentals and Limiting Processes. Int. J. Hydrogen Energy 2002, 27 (11-12), 1185–1193. https://doi.org/10.1016/S0360-3199(02)00131-3
Chen, J.; Wei, J.; Ma, C.; Yang, Z.; Li, Z.; Yang, X.; Wang, M.; Zhang, H.; Hu, J.; Zhang, C. Photosynthetic Bacteria-based Technology is a Potential Alternative to Meet Sustainable Wastewater Treatment Requirement? Environ. Int. 2020, 137, 105417. https://doi.org/10.1016/j.envint.2019.105417
[5] Lu, H.; Zhang, G.; Zheng, Z.; Meng, F.; Du, T.; He, S. Bio-conversion of Photosynthetic Bacteria from Non-toxic Wastewater to Realize Wastewater Treatment and Bioresource Recovery: A Review. Bioresour. Technol. 2019, 278, 383–399. https://doi.org/10.1016/j.biortech.2019.01.070
Imhoff, J. F. Systematics of Anoxygenic Phototrophic Bacteria. In Sulfur Metabolism in Photoorganisms; Hell, R., Dahl, C., Knaff, D., Leustek, T., Eds.; Springer: Dordrecht, 2008; pp 269–287. https://doi.org/10.1007/978-1-4020-6863-8_14
Ram, S.; Mitra, M.; Shah, F.; Tirkey, S. R.; Mishra, S. Bacteria as an Alternate Biofactory for Carotenoid Production: A Review of Its Applications, Opportunities and Challenges. J. Funct. Foods 2020, 67, 103867. https://doi.org/10.1016/j.jff.2020.103867
Wang, G.; Grammel, H.; Abou-Aisha, K.; Sägesser, R.; Ghosh, R. High-level Production of the Industrial Product Lycopene by the Photosynthetic Bacterium Rhodospirillum rubrum. Appl. Environ. Microbiol. 2012, 78 (20), 7205–7215. https://doi.org/10.1128/AEM.00545-12
Mapelli-Brahm, P.; Gómez-Villegas, P.; Gonda, M. L.; León-Vaz, A.; León, R.; Mildenberger, J.; Rebours, C.; Saravia, V.; Vero, S.; Vila, E.; Meléndez-Martínez, A. J. Microalgae, Seaweeds and Aquatic Bacteria, Archaea, and Yeasts: Sources of Carotenoids with Potential Antioxidant and Anti-inflammatory Health-promoting Actions in the Sustainability Era. Mar. Drugs 2023, 21 (6), 340. https://doi.org/10.3390/md21060340
Cohen, A. C.; Dichiara, E.; Jofré, V.; Antoniolli, A.; Bottini, R.; Piccoli, P. Carotenoid Profile Produced by Bacillus licheniformis RT4M10 Isolated from Grapevines Grown in High Altitude and Their Antioxidant Activity. Int. J. Food Sci. Technol. 2018, 53 (11), 2697–2705. https://doi.org/10.1111/ijfs.13879
Liu, L.; Li, J.; Cai, X.; Ai, Y.; Long, H.; Ren, W.; Huang, A.; Zhang, X.; Xie, Z. Dietary Supplementation of Astaxanthin is Superior to Its Combination with Lactococcus lactis in Improving the Growth Performance, Antioxidant Capacity, Immunity and Disease Resistance of White Shrimp (Litopenaeus vannamei). Aquacult. Rep. 2022, 24, 101124. https://doi.org/10.1016/j.aqrep.2022.101124
Yousefi, M.; Nedaei, S.; Farsani, M. N.; Ghafarifarsani, H.; Zhang, M.; Du, Z. Dietary Chrysin Supplementation Improves Growth Performance, Immune Responses, Antioxidant Status, and Resistance against Crowding Stress in Rainbow Trout. Aquacult. Rep. 2023, 32, 101708. https://doi.org/10.1016/j.aqrep.2023.101708
Niedzwiedzki, D. M.; Dilbeck, P. L.; Tang, Q.; Mothersole, D. J.; Martin, E. C.; Bocian, D. F.; Holten, D.; Hunter, C. N. Functional Characteristics of Spirilloxanthin and Keto-bearing Analogues in Light-harvesting LH2 Complexes from Rhodobacter sphaeroides with a Genetically Modified Carotenoid Synthesis Pathway. Biochim. Biophys. Acta, Bioenerg. 2015, 1847 (6-7), 640–655. https://doi.org/10.1016/j.bbabio.2015.04.001
Cahoon, L. B.; Halkides, C. J.; Song, B.; Williams, C. M.; Dubay, G. R.; Fries, A.; Farmer, J.; Fridrich, W.; Brookshire, C. Swine Waste as a Source of Natural Products: A Carotenoid Antioxidant. Agric. Sci. 2012, 3 (6), 806–815. https://doi.org/10.4236/as.2012.36098
Lee, S.; Lur, H.; Liu, C. From Lab to Farm: Elucidating the Beneficial Roles of Photosynthetic Bacteria in Sustainable Agriculture. Microorganisms 2021, 9 (12), 2453. https://doi.org/10.3390/microorganisms9122453
Chumpol, S.; Kantachote, D.; Nitoda, T.; Kanzaki, H. Administration of Purple Nonsulfur Bacteria as Single Cell Protein by Mixing with Shrimp Feed to Enhance Growth, Immune Response and Survival in White Shrimp (Litopenaeus vannamei) Cultivation. Aquaculture 2018, 489, 85–95. https://doi.org/10.1016/j.aquaculture.2018.02.009
Alloul, A.; Wille, M.; Lucenti, P.; Bossier, P.; Van Stappen, G.; Vlaeminck, S. E. Purple Bacteria as Added-value Protein Ingredient in Shrimp Feed: Penaeus vannamei Growth Performance, and Tolerance against Vibrio and Ammonia Stress. Aquaculture 2021, 530, 735788. https://doi.org/10.1016/j.aquaculture.2020.735788
Koga, A.; Goto, M.; Hayashi, S.; Yamamoto, S.; Miyasaka, H. Probiotic Effects of a Marine Purple Non-sulfur Bacterium, Rhodovulum sulfidophilum KKMI01, on Kuruma Shrimp (Marsupenaeus japonicus). Microorganisms 2022, 10 (2), 244. https://doi.org/10.3390/microorganisms10020244
Sudpraseart, C.; Shinn, A. P.; Pooljun, C.; Sirimanapong, W. Efficacy of Rhodobacter sphaeroides TISTR 1529 on the Growth Performance, Immune Response, and Amino Acid Profile of Pacific Whiteleg Shrimp, Penaeus vannamei. Aquaculture 2025, 599, 742081.
Smith, D.; Tabrett, S.; Barclay, M.; Irvin, S. The Efficacy of Ingredients Included in Shrimp Feeds to Stimulate Intake. Aquacult. Nutr. 2005, 11 (4), 263–272. https://doi.org/10.1111/j.1365-2095.2005.00349.x
Yuan, Y.; Lawrence, A. L.; Chehade, S. B.; Jensen, K. E.; Barry, R. J.; Fowler, L. A.; Makowsky, R.; Powell, M. L.; Watts, S. A. Feed Intake as an Estimation of Attractability in Pacific White Shrimp Litopenaeus vannamei. Aquaculture 2021, 532, 736041. https://doi.org/10.1016/j.aquaculture.2020.736041
Tantikitti, C. Feed Palatability and the Alternative Protein Sources in Shrimp Feed. Songklanakarin J. Sci. Technol. 2014, 36 (1), 51–55.
Al-Souti, A.; Gallardo, W.; Claereboudt, M.; Mahgoub, O. Attractability and Palatability of Formulated Diets Incorporated with Chicken Feather and Algal Meals for Juvenile Gilthead Seabream, Sparus aurata. Aquacult. Rep. 2019, 14, 100199. https://doi.org/10.1016/j.aqrep.2019.100199
Wang, C.; Chuprom, J.; Wang, Y.; Fu, L. Beneficial Bacteria for Aquaculture: Nutrition, Bacteriostasis and Immunoregulation. J. Appl. Microbiol. 2019, 128 (1), 28–40. https://doi.org/10.1111/jam.14383
Hamza, F.; Zinjarde, S. Use of Marine Microorganisms in Designing Anti-infective Strategies for Sustainable Aquaculture Production. J. Appl. Microbiol. 2023, 134 (6), lxad128. https://doi.org/10.1093/jambio/lxad128
Higuchi-Takeuchi, M.; Morisaki, K.; Numata, K. A Screening Method for the Isolation of Polyhydroxyalkanoate-producing Purple Non-sulfur Photosynthetic Bacteria from Natural Seawater. Front. Microbiol. 2016, 7, 1509. https://doi.org/10.3389/fmicb.2016.01509
Nithya, C.; Pandian, S. K. Isolation of Heterotrophic Bacteria from Palk Bay Sediments Showing Heavy Metal Tolerance and Antibiotic Production. Microbiol. Res. 2010, 165 (7), 578–593. https://doi.org/10.1016/j.micres.2009.10.004
Sibero, M. T.; Igarashi, Y.; Radjasa, O. K.; Sabdono, A.; Trianto, A.; Zilda, D. S.; Wijaya, Y. J. Sponge-associated Fungi from a Mangrove Habitat in Indonesia: Species Composition, Antimicrobial Activity, Enzyme Screening and Bioactive Profiling. Int. Aquat. Res. 2019, 11, 173–186. https://doi.org/10.1007/s40071-019-0227-8
Britton, G.; Liaaen-Jensen, S.; Pfender, H. Carotenoids: Handbook; Birkhäuser Verlag: Basel, 2004. https://doi.org/10.1007/978-3-0348-7836-4
Kiriratnikom, S. Evaluation of Possible Application of Photosynthetic Bacteria in Black Tiger Shrimp (Penaeus monodon). Ph.D. Thesis, Prince of Songkla University, Songkhla, Thailand, 2006.
Mukherjee, T.; Bose, S.; Mukhopadhyay, S. K. Antioxidant Properties of the Carotenoid Extracts of Three Deinococcus-Thermus Phylum Bacteria, Meiothermus sp. Strains RP and TP and Thermus sp. Strain YY from Paniphala Hot Spring, India. Nutrire 2017, 42, 1–11. https://doi.org/10.1186/s41110-017-0032-3
Fang, C.; Ku, K.; Lee, M.; Su, N. Influence of Nutritive Factors on C50 Carotenoids Production by Haloferax mediterranei ATCC 33500 with Two-stage Cultivation. Bioresour. Technol. 2010, 101 (16), 6487–6493. https://doi.org/10.1016/j.biortech.2010.03.044
Kim, S.; Jeon, H.; Bai, S. C.; Kim, K.; Lee, S.; Hur, J. W.; Han, H. Effects of Dietary Supplementation with Arthrobacter bussei Powder on Growth Performance, Antioxidant Capacity, and Innate Immunity of Pacific White Shrimp (Litopenaeus vannamei). Aquacult. Rep. 2022, 25*, 101270. https://doi.org/10.1016/j.aqrep.2022.101270
Suresh, A. V.; Vasagam, K. P. K.; Nates, S. Attractability and Palatability of Protein Ingredients of Aquatic and Terrestrial Animal Origin, and Their Practical Value for Blue Shrimp, Litopenaeus stylirostris Fed Diets Formulated with High Levels of Poultry Byproduct Meal. Aquaculture 2011, 319 (1-2), 132–140. https://doi.org/10.1016/j.aquaculture.2011.06.039
Chirapongsatonkul, N.; Srichanun, M.; U-taynapun, K. The Impact of a Mixture of Biofloc Fermentation Medium and Vinasse on Attractability, Palatability, and Antibacterial Properties against Multi-antibiotic Resistant Aeromonas veronii. Int. J. Agric. Technol. 2019, 15 (6), 845–858.
U-taynapun, K.; Nganwisuthiphan, T.; Chirapongsatonkul, N. Species Diversity and Existence of Virulence Genes in Clinical Aeromonas spp. Causing Motile Aeromonas Septicemia (MAS) Isolated from Cultured Nile Tilapia (Oreochromis niloticus). Int. J. Agric. Technol. 2020, 16 (4), 749–760.
Tamura, K.; Stecher, G.; Kumar, S. MEGA11: Molecular Evolutionary Genetics Analysis Version 11. Mol. Biol. Evol. 2021, 38 (7), 3022–3027. https://doi.org/10.1093/molbev/msab120
Maresca, J. A.; Romberger, S. P.; Bryant, D. A. Isorenieratene Biosynthesis in Green Sulfur Bacteria Requires the Cooperative Actions of Two Carotenoid Cyclases. J. Bacteriol. 2008, 190 (19), 6384–6391. https://doi.org/10.1128/JB.00758-08
Vila, E.; Hornero-Méndez, D.; Azziz, G.; Lareo, C.; Saravia, V. Carotenoids from Heterotrophic Bacteria Isolated from Fildes Peninsula, King George Island, Antarctica. Biotechnol. Rep. 2019, 21, e00306. https://doi.org/10.1016/j.btre.2019.e00306
Sutherland, G. A.; Qian, P.; Hunter, C. N. Swainsbury, D. J.; Hitchcock, A. Engineering Purple Bacterial Carotenoid Biosynthesis to Study the Roles of Carotenoids in Light-harvesting Complexes. Methods Enzymol. 2022, 674, 137–184. https://doi.org/10.1016/bs.mie.2022.04.001
Kim, J. K.; Lee, B. Mass Production of Rhodopseudomonas palustris as Diet for Aquaculture. Aquacult. Eng. 2000, 23 (4), 281–293. https://doi.org/10.1016/S0144-8609(00)00057-1
Soon, T. K.; Al-Azad, S.; Ransangan, J. Isolation and Characterization of Purple Non-sulfur Bacteria, Afifella marina, Producing Large Amount of Carotenoids from Mangrove Microhabitats. J. Microbiol. Biotechnol. 2014, 24 (8), 1034–1043. https://doi.org/10.4014/jmb.1308.08072
López, G.-D.; Álvarez-Rivera, G.; Carazzone, C.; Ibáñez, E.; Leidy, C.; Cifuentes, A. Bacterial Carotenoids: Extraction, Characterization, and Applications. Crit. Rev. Anal. Chem. 2023, 53 (6), 1239–1262. https://doi.org/10.1080/10408347.2021.2016366
Gao, Y.; Focsan, A. L.; Kispert, L. D. Antioxidant Activity in Supramolecular Carotenoid Complexes Favored by Nonpolar Environment and Disfavored by Hydrogen Bonding. Antioxidants 2020, 9 (7), 625. https://doi.org/10.3390/antiox9070625
Imhoff, J. F. Rhodocista. In Bergey's Manual of Systematics of Archaea and Bacteria; Wiley: 2015; pp 1–5. https://doi.org/10.1002/9781118960608.gbm00895
Dey, A.; Srivastava, N.; Sharma, S.; Verma, V.; Rao, L.; Chaudhary, M.; Jain, S.; Pinapati, K. K. Neuroprotective Effects of Carotenoid Rich Verbesina encelioides Flower Extract in Scopolamine Induced Memory Impaired Rats Mimicking Alzheimer's Disease. Preprints 2024, 2024031767. https://doi.org/10.20944/preprints202403.1767.v1
Wu, G.-S.; Chung, Y.-M.; Lin, W.-Y.; Chen, S.-Y.; Huang, C.-H. Effect of Substituting De-hulled or Fermented Soybean Meal for Fish Meal in Diets on Growth of Hybrid Tilapia, Oreochromis niloticus and O. aureus. J. Fish. Soc. Taiwan 2003, 30 (4), 291–297.
Bardera, G.; Owen, M. A.; Façanha, F. N.; Alcaraz-Calero, J. M.; Sloman, K. A.; Alexander, M. E. Assessing Feed Attractability in Pacific White Shrimp (Litopenaeus vannamei) Using an Automated Tracking Software. Aquaculture 2020, 529, 735692. https://doi.org/10.1016/j.aquaculture.2020.735692
Lim, L.; Liew, K.; Ebi, I.; Shapawi, R.; Lal, M. T. M.; Liew, H. J.; Hamasaki, K.; Masuda, R.; Kawamura, G. Amino Acids as Chemoattractant and Feeding Stimulant for the Commercially Farmed Decapod Crustaceans: A Brief Review. Aquacult. Res. 2022, 53 (2), 333–343. https://doi.org/10.1111/are.15591
Archadale, M. V.; Nakamura, K. Responses to the Swimming Crab Portunus pelagicus to Amino Acids and Mono-and Disaccharides. Nippon Suisan Gakkaishi 1992, 58 (2), 165–172. https://doi.org/10.2331/suisan.58.165
Patthawaro, S.; Saejung, C. Production of Single Cell Protein from Manure as Animal Feed by Using Photosynthetic Bacteria. MicrobiologyOpen 2019, 8 (10), e913. https://doi.org/10.1002/mbo3.913