Impact of Marine Fish Amino Acid on Yield Parameters and Preventive Antioxidant in Okra
Main Article Content
Abstract
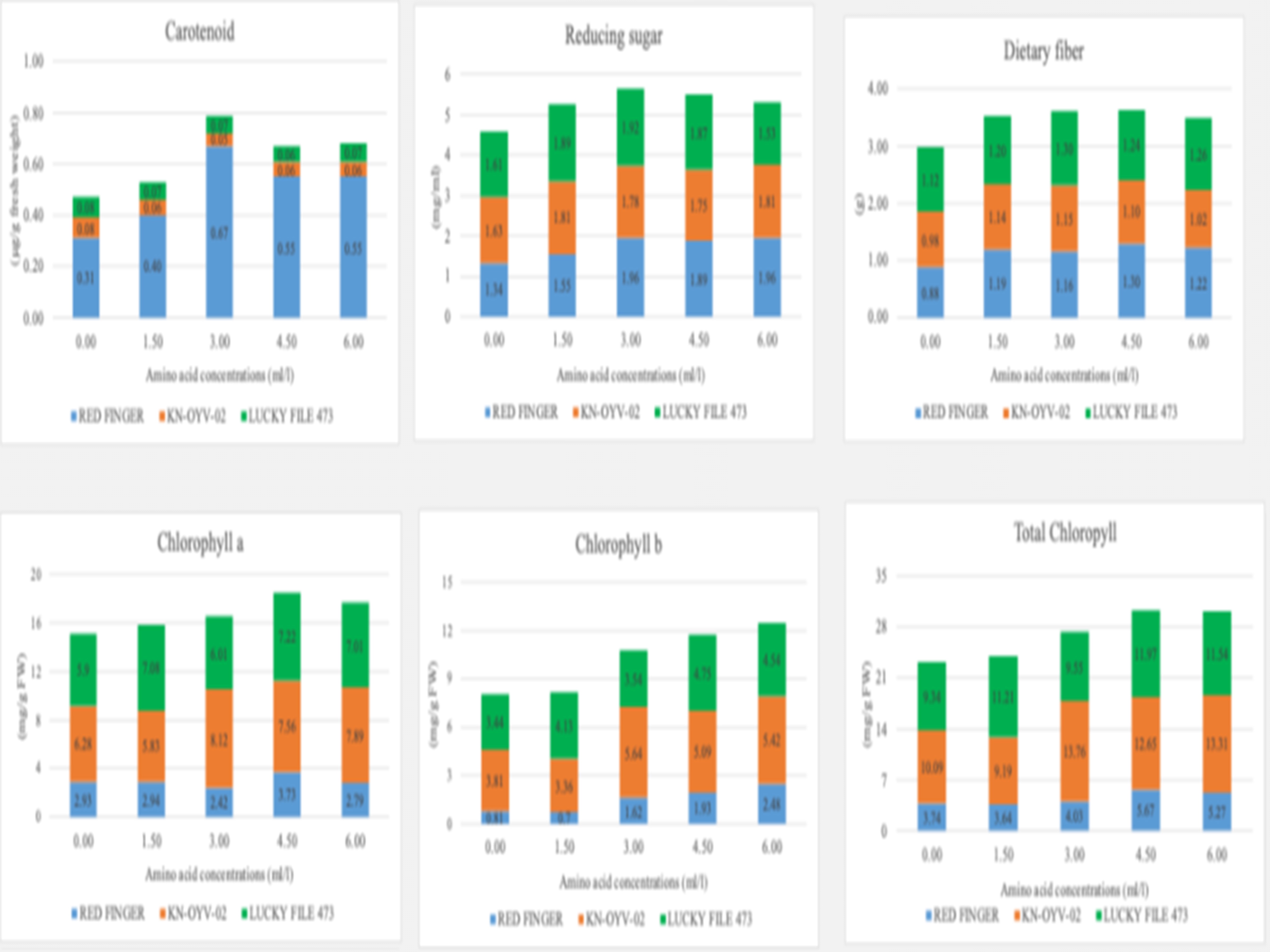
A field experiment was carried out to evaluate the effects of foliar-applied marine fish amino acids (MFA) on the growth, yield, yield components, and preventive antioxidant capacity of okra, as well as the interaction between okra varieties and MFA concentrations. The study used a split-plot design with four replications. Three okra varieties — RED FINGER, KN–OYV–02, and LUCKY FILE 473 — were assigned to main plots. Five concentrations were tested in the subplots (0.00, 1.50, 3.00, 4.50, and 6.00 ml/l). Yield per plant — a key indicator for growers — did not differ significantly among the three varieties but responded to MFA levels-plants treated with 3.00, 4.50, or 6.00 ml/l. MFA showed no significant differences among these higher concentrations; however, all produced higher yields than the untreated control and the 1.50 ml/l treatment. The greatest yield (1,271.49 g/plant) was obtained at 3.00 ml/l, followed by 4.50 ml/l (1,251.22 g/plant) and 6.00 ml/l (1,215.51 g/plant). Reducing sugar content did not vary significantly among the okra varieties but was influenced by MFA levels. LUCKY FILE 473 recorded the highest reducing sugar (~1.77 mg/ml). The concentrations 3.00, 4.50, and 6.00 ml/l yielded the highest reducing sugar levels (1.89, 1.84, and 1.77 mg/ml, respectively), with no significant difference among them, while the control plants had the lowest value (1.53 mg/ml). Among pigment traits, RED FINGER exhibited the lowest chlorophyll a, chlorophyll b, and total chlorophyll contents (2.96, 1.51, and 4.47 mg/g FW, respectively) but had the highest carotenoid concentration (0.50 µg/g FW).
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
References
Benchasri, S.; Pruthikanee, P. Genetic Variability for Yield and Yield Components of Thai Chilli (Capsicum spp.) Landraces under Inorganic and Organic Agricultural Systems. Aust. J. Crop Sci. 2018, 12, 126–134. https://doi.org/10.21475/ajcs.18.12.01.pne778
Liu, J.; Lin, X.; Wang, X.; Feng, L.; Zhu, S.; Tian, R.; Fang, J.; Tao, A.; Fang, P.; Qi, J.; Zhang, L.; Huang, Y.; Xu, J. Genomic and Cytogenetic Analyses Reveal Satellite Repeat Signature in Allotetraploid Okra (Abelmoschus esculentus). BMC Plant Biol. 2024, 24, 71. https://doi.org/10.1186/s12870-024-04739-9
Benchasri, S. Okra (Abelmoschus esculentus (L.) Moench) as a Valuable Vegetable of the World. Field Veg. Crop Res. 2012, 49, 105–112. https://doi.org/10.5937/ratpov49-1172
Dantas, T. L.; Buriti, F. C. A.; Florentino, E. R. Okra (Abelmoschus esculentus L.) as a Potential Functional Food Source of Mucilage and Bioactive Compounds with Technological Applications and Health Benefits. Plants 2021, 10 (8), 1683. https://doi.org/10.3390/plants10081683
Hamdan, Y. A. S.; Hawamda, A. I. M.; Salimia, R. B.; Salman, M. Genetic Diversity Assessment of Palestinian Okra Landraces (Abelmoschus esculentus L.) through RAPD Marker. Genet. Resour. Crop Evol. 2024, in press. https://doi.org/10.21203/rs.3.rs-3536240/v1
Benchasri, S.; Simla, S.; Harakotr, B. The Effect of Genotypic Variability on the Yield and Yield Components of Okra (Abelmoschus esculentus L. Moench) in Thailand. Asian J. Agric. Biol. 2020, 8(4), 480–490.
Sun, J.; Xu, G.; Hu, Y.; Zheng, Y.; Wang, X.; Yang, J.; Yang, M.; Xie, D.; Dai, Z. Genome-Wide Assessment of Genetic Diversity and Association Mapping for Salt Tolerance Traits in Okra (Abelmoschus esculentus L. Moench) Using Genotyping-by-Sequencing. Sci. Hortic. 2023, 313, 111922. https://doi.org/10.1016/j.scienta.2023.111922
Coban, S. N.; Polatoglu, I.; Eroglu, E. M. Methyl Cellulose/Okra Mucilage Composite Films, Functionalized with Hypericum perforatum Oil and Gentamicin, as a Potential Wound Dressing. Int. J. Biol. Macromol. 2024, 251 (1), 127757. https://doi.org/10.1016/j.ijbiomac.2023.127757
Graham, J. O.; Agbenorhevi, J. K.; Kpodo, F. M. Total Phenol Content and Antioxidant Activity of Okra Seeds from Different Genotypes. Am. J. Food Nutr. 2015, 5, 90–94. https://doi.org/10.12691/ajfn-5-3-2
Zhang, T.; Xiang, J.; Zheng, G.; Yan, R.; Min, X. Preliminary Characterization and Antihyperglycemic Activity of a Pectic Polysaccharide from Okra (Abelmoschus esculentus (L.) Moench). J. Funct. Foods 2018, 41, 19–24. https://doi.org/10.1016/j.jff.2017.12.028
Elkhalifa, A. E.; Alshammari, E.; Adnan, M.; Alcantara, J. C.; Awadelkareem, A. M.; Eltoum, N. E.; Mehmood, K.; Panda, B. P.; Ashraf, S. A. Okra (Abelmoschus esculentus) as a Potential Dietary Medicine with Nutraceutical Importance for Sustainable Health Applications. Molecules 2021, 26(3), 696. https://doi.org/10.3390/molecules26030696
Jiang, N.; Liu, C.; Li, D.; Zhang, Z.; Liu, C.; Wang, D.; Niu, L.; Zhang, M. Evaluation of Freeze Drying Combined with Microwave Vacuum Drying for Functional Okra Snacks: Antioxidant Properties, Sensory Quality, and Energy Consumption. LWT–Food Sci. Technol. 2017, 82, 216–226. https://doi.org/10.1016/j.lwt.2017.04.015
Hildebrandt, T. M.; et al. Amino Acid Catabolism in Plants. Mol. Plant 2015, 8(11), 1563–1579. https://doi.org/10.1016/j.molp.2015.09.005
Sarojnee, D. Y.; Navindra, B.; Chandrabose, S. Effect of Naturally Occurring Amino Acid Stimulants on the Growth and Yield of Hot Peppers (Capsicum annuum L.). J. Anim. Plant Sci. 2009, 5(1), 414–424.
Rebecca, L. J.; Sharmila, S.; Paul Das, M.; Seshiah, C. Extraction and Purification of Carotenoids from Vegetables. J. Chem. Pharm. Res. 2014, 6 (4), 594–598.
Giusti, M. M.; Wrolstad, R. E. Characterization and Measurement of Anthocyanins by UV–Visible Spectroscopy. Curr. Protoc. Food Anal. Chem. 2001, F1.2.1–F1.2.13. https://doi.org/10.1002/0471142913.faf0102s00
Somogyi, M. Notes on Sugar Determination. J. Biol. Chem. 1952, 195, 19–23. https://doi.org/10.1016/S0021-9258(19)50870-5
Andrade, N. J. P.; Monteros-Altamirano, A.; Bastidas, C. G. T.; Sørensen, M. Morphological, Sensorial and Chemical Characterization of Chilli Peppers (Capsicum spp.) from the CATIE Genebank. Agronomy 2020, 10 (11), 1732. https://doi.org/10.3390/agronomy10111732
Karem, M. H.; Haidery, A. A. A. Induced Systemic Resistance of Okra (Abelmoschus esculentus L.) against Okra Yellow Vein Mosaic Virus Using Amino Acid and Algae Extracts. Pak. J. Phytopathol. 2022, 34(2), 213–220. https://doi.org/10.33866/phytopathol.034.02.0799
Han, G.; Li, Y.; Yang, Z.; Wang, C.; Zhang, Y.; Wang, B. Molecular Mechanisms of Plant Trichome Development. Front. Plant Sci. 2022, 13, 910228. https://doi.org/10.3389/fpls.2022.910228
Khan, S.; Yu, H.; Li, Q.; Gao, Y.; Sallam, B. N.; Wang, S.; Liu, P.; Jiang, W. Exogenous Application of Amino Acids Improves the Growth and Yield of Lettuce by Enhancing Photosynthetic Assimilation and Nutrient Availability. Agronomy 2019, 9, 266. https://doi.org/10.3390/agronomy9050266
Shekari, G.; Javanmardi, J. Effects of Foliar Application of Pure Amino Acid and Amino Acid-Containing Fertilizer on Broccoli (Brassica oleracea L. var. italica) Transplant. Adv. Crop Sci. Technol. 2017, 5, 280. https://doi.org/10.4172/2329-8863.1000280
Seyedi, A.; Fathi, S.; Movlodzadeh, R. The Effect of Biostimulants Based on Free Amino Acids on Some Growth and Physiological Parameters of Dracocephalum moldavica L. under Salinity Stress. J. Med. Plants By-Prod. 2023, in press.
Krishnamoorthy, R.; Alshatwi, A. A.; Subbarayan, S.; Bharathi. Impact of Farm-Made Liquid Organic Nutrients Jevamirtham and Fish Amino Acid on Growth and Nutritional Status in Different Seasons of Abelmoschus esculentus: A Self-Sustainable Field Trial. Org. Agric. 2019, 9(3–4), 333–344. https://doi.org/10.1007/s13165-018-0205-2
Koukounaras, A.; Tsouvaltzis, P.; Siomos, A. S. Effect of Root and Foliar Application of Amino Acids on the Growth and Yield of Greenhouse Tomato under Different Fertilization Levels. J. Food Agric. Environ. 2013, 11, 644–648.
Teixeira, W. F.; Fagan, E. B.; Soares, L. H.; Umburanas, R. C.; Reichardt, K.; Neto, D. D. Foliar and Seed Application of Amino Acids Affects the Antioxidant Metabolism of the Soybean Crop. Front. Plant Sci. 2017, 8, 327. https://doi.org/10.3389/fpls.2017.00327
Baqir, A.; Naqeeb, A. Effect of Some Amino Acids on Tillering and Yield of Three Bread Wheat Cultivars. Iraqi J. Agric. Sci. 2019, 50, 20–30. https://doi.org/10.36103/ijas.v50iSpecial.173
Ijarotimi, O. S.; Akinola-Ige, A. O.; Oluwajuyitan, T. D. Okra Seed Proteins: Amino Acid Profile, Free Radical Scavenging Activities and Inhibition of Diabetes- and Hypertension-Converting Enzyme Indices. Measurement: Food 2023, 11, 100101. https://doi.org/10.1016/j.meafoo.2023.100101