Effect of Plant Growth Regulators on In Vitro Shoot and Root Induction and Acclimatization of Gymnocalycium mihanovichii LB2178 Agua Dulce (Cactaceae)
Main Article Content
Abstract
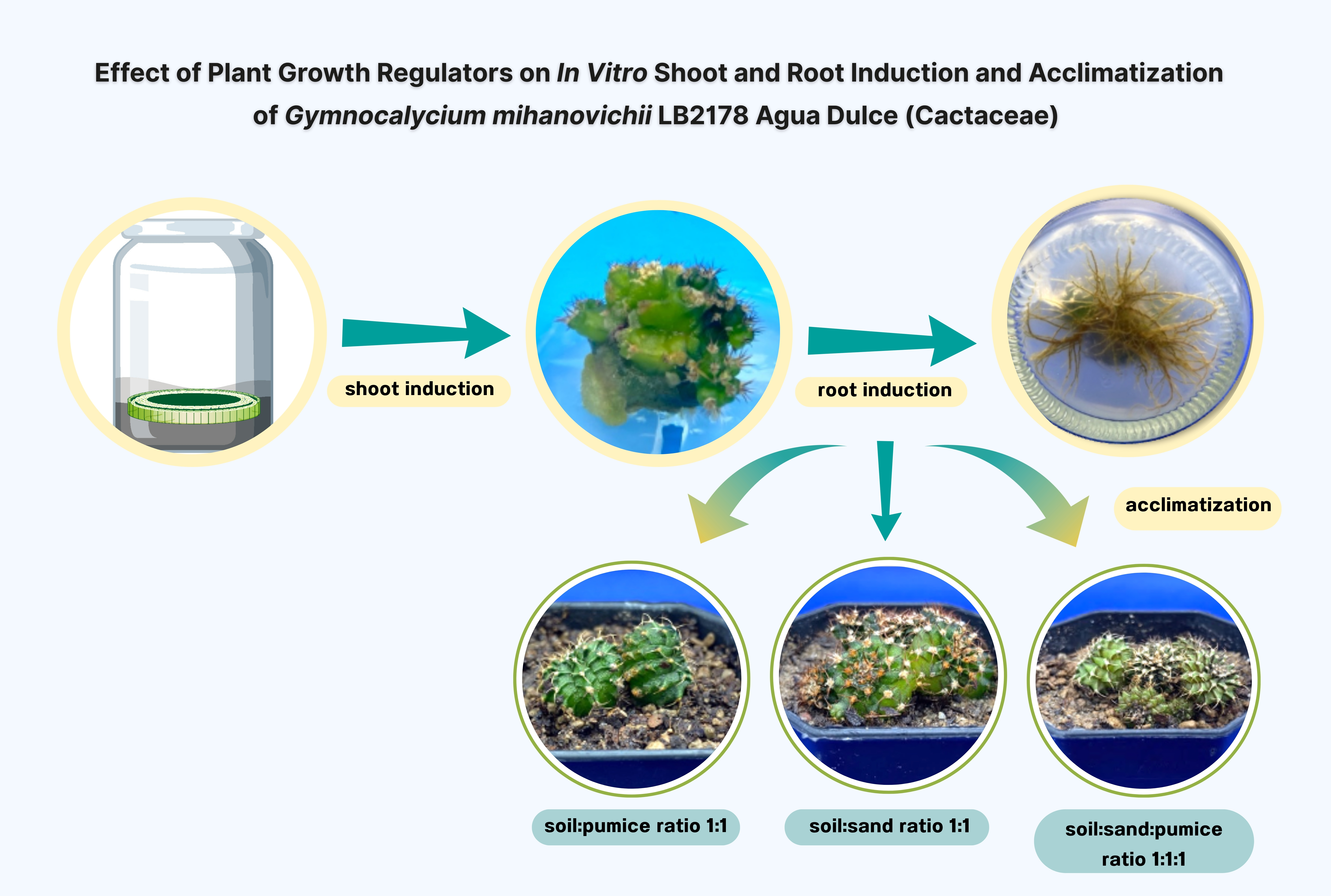
Gymnocalycium mihanovichii LB2178 Agua Dulce is a popular cactus still in demand, known for its distinctive shape and ease of growth. It can be propagated by tissue culture to produce strong, disease-free plants. Therefore, the aim of this research was 1) to study the effect of BA and IBA hormones and types of explants on the growth rate of G. mihanovichii LB2178 Agua Dulce cactus. and 2) to study suitable planting materials for survival and growth of G. mihanovichii LB2178 Agua Dulce cactus under natural conditions. After 3 months of culture, the results showed that the explant sectioned longitudinally were cultured on MS medium supplemented with 0 and 2 mg/L BA gave the highest survival, 100%, and shoot induction, 86.67%, the explant sectioned transversally were cultured on MS medium supplemented with 1 mg/L BA gave the highest number of shoot, 4.20 shoots/explant, and shoot length, 0.36 mm respectively, significant difference at p≤0.05. Following three months of cultivation, the findings indicated that shoot explants cultured on MS medium supplemented with 1 mg/L IBA showed the highest root induction (91.67%), average number of roots (20.47 roots/explant), and average root length (7.63 mm), with significant differences at p≤0.05. After 3 months, the rooted plants were removed from the tissue culture room and transferred to sterile plastic pots. The result showed that G. mihanovichii LB2178 Agua Dulce grown in potting soil: sand, showed the highest survival rate of 100%.
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
References
Hernández-Hernández, T.; Hernández, H. M.; De-Nova, J. A.; Puente, R.; Eguiarte, L. E.; Magallón, S. Phylogenetic Relationships and Evolution of Growth Form in Cactaceae (Caryophyllales, Eudicotyledoneae). Am. J. Bot. 2011, 98, 44–61. https://doi.org/10.3732/ajb.1000129
Goettsch, B.; Hilton-Taylor, C.; Cruz-Piñón, G.; et al. High Proportion of Cactus Species Threatened with Extinction. Nat. Plants 2015, 1, 1–7. https://doi.org/10.1038/nplants.2015.142
Boonyang, S. Business Plan for Building a New Business SB Cactus Garden. In Small and Medium-Sized Enterprises; Graduate School, Bangkok University: Bangkok, Thailand, 2016; pp 15–21. (in Thai)
Brutsch, M. O.; Scott, M. B. Extending the Fruiting Season of Spineless Prickly Pear (Opuntia ficus-indica). J. S. Afr. Soc. Hortic. Sci. 1991, 1, 73–76. https://doi.org/10.17660/ActaHortic.2009.811.7
Cota-Sánchez, J. H.; Crouch, D. S. Notes on the Floral Biology of Praecereus euchlorus subsp. euchlorus (Cactaceae). Schumannia 2008, 5, 98–103.
Heywood, V. Conservation and Sustainable Use of Wild Species as Sources of New Ornamentals. In Int. Symp. Sustainable Use of Plant Biodiversity; 2001; Vol. 598, pp 43–53.
Walters, M.; Figueiredo, E.; Crouch, N. R.; Winter, P. J. D.; Smith, G. F.; Zimmermann, H. G.; Mashope, B. K. Naturalised and Invasive Succulents of Southern Africa. Abc Taxa 2011, 11, 91–100.
Imraporn, S. Carry Out a Garden with a New Hybrid Cactus Makeover to Please Customers. W. Khaenket 2016, 40, 191–194. (in Thai)
Rittirong, C.; Keereeped, S.; Rotduang, P.; Ramasoot, S. Effect of Plant Growth Regulators on Anthocyanin Production from Shoot Culture of Gymnocalycium damsii. Wichcha J. 2020, 39, 116–128. (in Thai)
Gutiérrez-Quintana, L.; Zúñiga-Rizo, C.; Burgos, A.; Portillo, L. Micropropagation of Prickly Pear by Axillary Shoot Proliferation. Bio-Protocol 2018, 8, e2923.
Shehu, U. I.; Sani, L. A.; Ibrahim, A. B. Auxin-Induced Rooting of Cactus Pear (Opuntia ficus-indica) Cladodes for Rapid On-Farm Propagation. Afr. J. Agric. Res. 2016, 11, 898–900.
George, E. F.; Hall, M. A.; De Klerk, G.-J. Plant Tissue Culture Procedure—Background. In Plant Propagation by Tissue Culture, 3rd ed.; Springer: Dordrecht, 2008; Vol. 1, pp 1–28. https://doi.org/10.1007/s11240-008-9357-1
Bonga, J. M. Can Explant Choice Help Resolve Recalcitrance Problems in In Vitro Propagation? Trees 2017, 31, 781–789. https://doi.org/10.1007/s00468-016-1509-z
Rocha, D. I.; Monte-Bello, C. C.; Dornelas, M. C. Alternative Induction of de Novo Shoot Organogenesis or Somatic Embryogenesis. Plant Cell Tissue Organ Cult. 2014. https://doi.org/10.1007/s11240-014-0663-5
Torres-Silva, G.; Resende, S. V.; Lima-Brito, A.; Bezerra, H. B.; Santana, J. R. F.; Schnadelbach, A. S. In Vitro Shoot Production and Genetic Instability of Melocactus glaucescens. S. Afr. J. Bot. 2018, 115, 100–107. https://doi.org/10.1016/j.sajb.2018.01.001
Masakee, N.; Yenchon, S.; Te-chato, S. Effect of Plant Growth Regulators on In Vitro Callus and Shoot Induction of Haworthia. Songklanakarin J. Plant Sci. 2016, 3, 76–82. (in Thai)
Marhri, A.; Tikent, A.; Garros, L.; Merah, O.; Elamrani, A.; Hano, C.; Abid, M.; Addi, M. Rapid and Efficient In Vitro Propagation Protocol. Horticulturae 2023, 9, 491. https://doi.org/10.3390/horticulturae9040491
Leva, A. Innovative Protocol for “Ex Vitro Rooting” on Olive Micropropagation. Cent. Eur. J. Biol. 2011, 6, 352–358. https://doi.org/10.2478/s11535-011-0010-3
Santos-Díaz, M. d.; Alvarado-Rodríguez, J. In Vitro Regeneration of Turbincarpus mombergeri. Plant Cell Tissue Organ Cult. 2022, 148, 271–279. https://doi.org/10.1007/s11240-021-02181-5
Huong, T. T.; Tran, T. T. Efficient In Vitro Plantlet Regeneration from Leaf Explant of Haworthia retusa. J. Hortic. Sci. 2023, 18, 181–188. https://doi.org/10.24154/jhs.v18i1.2161
Goveas, S. W.; Nivas, S. K.; Souza, L. D. Development of High Frequency Multiple Shoots in Yellow Cactus. J. Appl. Hortic. 2011, 13, 42–43. https://doi.org/10.37855/jah.2011.v13i01.09
Trinh, H. T.; Tran, T. T. Efficient In Vitro Plantlet Regeneration from Leaf Explant of Haworthia retusa. J. Hortic. Sci. 2023, 18, 181–188. https://doi.org/10.24154/jhs.v18i1.2161
Sawardekar, S.; Sherkar, S.; More, V.; Pawar, S.; Kokare, K. In Vitro Propagation of Valuable Succulents: A Review. Int. J. Multidiscip. Res. 2023, 5, 1–17. https://doi.org/10.36948/ijfmr.2023.v05i05.7931
Read, P.; Preece, J. Cloning: Plants—Micropropagation/Tissue Culture. In Encyclopedia of Agriculture and Food Systems; 2014; Vol. 2, pp 317–333. https://doi.org/10.1016/B978-0-444-52512-3.00224-2
Karthik, P. M.; Samydurai, P.; Subbaiyan, B.; Thangapandian, V.; Binu, T. In Vitro Propagation of Caralluma diffusa. Res. Plant Biol. 2013, 3, 8–17.
Kanchanakul, S. Roiphan Phrueksa Cactus; Amarin Printing and Publishing: Bangkok, 2014; pp 105–108. (in Thai)